Myelofibrosis (MF) is a myeloproliferative stem cell disorder arising from various clonal mutations in early myeloid-committed hematopoietic progenitor cells. The subsequent acquisition of additional mutations and cytokine signaling then heralds the recruitment of fibroblasts to the marrow space, leading to collagen deposition. MF disease behavior varies widely and hematopoietic stem cell transplantation remains the only curative therapeutic option. In non-transplant candidates, managing cytopenic disease is uniquely challenging as patients develop resistance to commonly-utilized Jak-inhibitors. Expanding the molecular landscape of the disease beyond Jak/Stat signaling with the goal of identifying new therapeutic targets has, accordingly, been an area of growing interest in MF research. Recently-published studies, including results from our own institution, have identified mutations that potentially activate Ras signaling as being associated with features of advanced MF (higher somatic mutation burden) as well as with shorter overall survival and progression to AML. The goals of our study is to expand on this prior data, characterize the activity of Ras signaling and determine its role in patients with MF.
Peripheral blood mononuclear cells (PBMCs) from 27 patients with myelofibrosis (MF) at a single institution underwent integrated genomic-transcriptomic sequencing analyses using the University of Michigan MiOncoSeq platform. PBMCs were separated by Ficoll® followed by cell lysis and protein quantification. Ras activity was measured by the binding of effector Ras binding domain (RBD) via ELISA. Patients with MF were compared to healthy donors and to patients with either polycythemia vera (PV) or essential thrombocythemia (ET). Ras activity in MF was examined across different mutations and with respect to various clinical parameters. Multivariate analyses were performed to assess the impact of each individual mutation on Ras activity. Accompanying statistical analyses for all assessments were conducted using two-way unpaired t-tests.
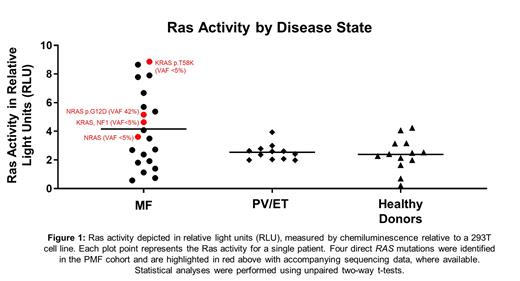
Overall, Ras activity in 21 assessable patients with MF was 1.64 fold higher than in PV/ET (12 patients, p=0.0462) and 1.74 fold higher than healthy donors (13 volunteers, p=0.0304). While the majority of patients in the MF cohort had driver mutations, including in JAK2 (73%) and CALR (15.4%), the individual types of driver mutations did not impact Ras activity. Within the Ras/MAP Kinase signaling pathway, mutations were present at a range of variant allele frequencies (VAF), including 2 patients with NRAS (one with VAF 42%, one <5%), 2 KRAS (both <5%), 2 NF1, 1 CBL and 1 RAF1 (VAFs not assessed). The individual impact of various driver mutations, epigenetic regulators and splicing factors as assessed by multivariate analysis identified a trend toward enhanced Ras activity with mutations in RAS, CALR, ASXL1, TET2 and TP53, suggesting that in addition to RAS, other mutations also activate Ras signaling in MF. With regards to clinical parameters, Ras activity was weakly correlated with spleen size and high molecular risk mutation (HMR) presence, although this did not reach statistical significance in this cohort due to a relatively small sample size. Next, we performed a differential enrichment of myeloid and lymphoid cells from PBMCs using EasySep™ and measured Ras activity in lymphoid and myeloid enriched cells. We observed that the ratio of myeloid:lymphoid activity in patients with MF with RAS/MAP Kinase pathway mutations is 3.8 times higher than in patients without such mutations (p=0.0012), suggesting that RAS pathway mutations have a predominant role in myeloid cells.
Our results show that the majority of patients with MF have hyperactive Ras signaling compared to PV/ET or healthy donors, regardless of the presence of alterations in the Ras signaling pathway. This underscores the importance of functional assessment of Ras activity in patients with MF and provides evidence that targeting Ras signaling in MF may lead to therapeutic benefit. We are currently expanding our analysis to a larger cohort of MF patients and performing preclinical testing of Ras/MAP Kinase pathway inhibitors as a means of overcoming Jak-inhibitor resistance.
Disclosures
Pettit:Protagonist Therapeutics, Inc.: Consultancy, Research Funding; Merck: Research Funding, Speakers Bureau; AbbVie: Consultancy, Research Funding. Talpaz:Sumitomo: Research Funding; Novartis: Research Funding; Morphosys: Research Funding; BMS: Other: Advisory Board Member; Sumitomo: Other: Advisory Board Member; BMS: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal